Categories
Latest blog
Tags
When sourcing intravenous access devices, selecting the right IV cannula supplier is essential for ensuring consistent quality and regulatory compliance.
Although the two terms are often used interchangeably in clinical settings, they are not exactly the same. For hospital purchasing departments, medical distributors, and OEM buyers, knowing these distinctions helps ensure correct product selection, inventory planning, and tender documentation.
This guide explains:
- The exact difference between IV cannula and IV catheter
- Clinical applications and size standards
- Material and safety considerations
- How to choose the right product for bulk procurement
- Key factors when selecting a reliable manufacturer
| Feature | IV Cannula | IV Catheter |
|---|---|---|
| Definition | A peripheral intravenous access device inserted into a vein | A broader term for flexible tubes inserted into the body |
| Common Clinical Use | IV fluid infusion, medication administration, blood transfusion | Urinary drainage, central venous access, arterial access, etc. |
| Typical Location | Peripheral veins (hand, forearm) | Depends on type (urinary, central, arterial, etc.) |
| Size Range | 14G–24G (color-coded) | Varies by application |
| Materials | FEP, PTFE, PU | Silicone, latex, PU, PTFE |
| Primary Buyers | Hospitals, emergency departments, distributors | Hospitals, specialty clinics |
| Wholesale MOQ | Commonly 10,000–50,000 pcs | Depends on catheter type |
Key takeaway:
An IV cannula is a specific type of peripheral IV catheter used for intravenous therapy.
Looking for a CE Certified IV Cannula Manufacturer?
✔ 14G–24G Available
✔ Safety & Standard Options
✔ OEM Private Label
✔ Bulk Pricing
View Product Specifications
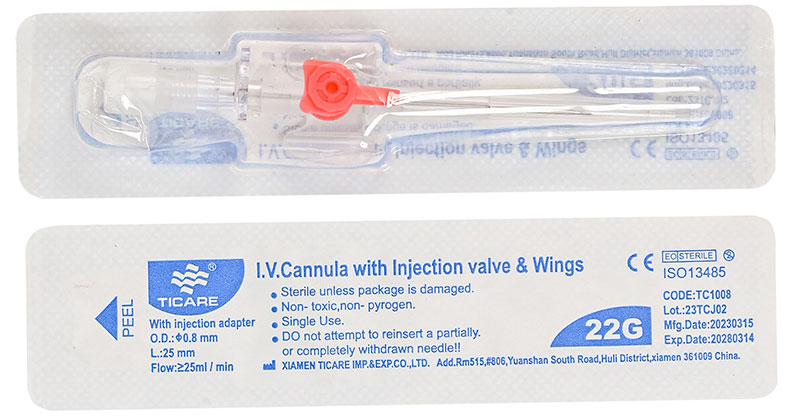
An IV cannula is a small, flexible tube inserted into a peripheral vein to deliver:
- Intravenous fluids
- Medications
- Blood products
- Nutritional solutions
It consists of:
- Stainless steel introducer needle
- Flexible catheter tube
- Injection port
- Luer lock fitting
- Protective cap
After insertion, the needle is removed, leaving only the flexible catheter in the vein.
| Gauge | Color | Typical Application |
|---|---|---|
| 14G | Orange | Trauma, rapid transfusion |
| 16G | Grey | Surgery, emergency |
| 18G | Green | Blood transfusion |
| 20G | Pink | General adult infusion |
| 22G | Blue | Elderly, fragile veins |
| 24G | Yellow | Pediatric patients |
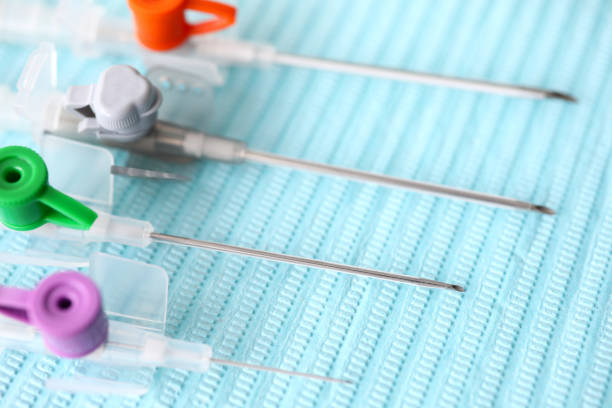
For hospital procurement, selecting the correct size distribution mix is critical for inventory efficiency.
An IV catheter is a broader category of flexible medical tubing inserted into the body for fluid administration or drainage.
Types include:
- Peripheral IV catheter (IV cannula)
- Central venous catheter (CVC)
- Peripherally inserted central catheter (PICC)
- Arterial catheter
- Urinary catheter
In daily hospital usage, “IV catheter” often refers to a peripheral IV cannula. However, in regulatory and procurement documents, the terminology must be precise.
From a purchasing perspective, the key differences include:
IV cannula = Peripheral intravenous access only
IV catheter = Multiple catheter categories
Misclassification in tenders may cause compliance issues.
Depending on region:
- CE (EU MDR)
- FDA 510(k) (USA)
- ISO 13485 manufacturing compliance
Safety IV cannulas may be mandatory in certain markets (e.g., EU hospitals to prevent needlestick injuries).
Hospitals increasingly require:
- Passive safety IV cannula
- Active safety IV cannula
- Needle shield protection
Safety versions typically cost more but reduce occupational injury risk.
For distributors, offering both standard and safety models expands market coverage.
When purchasing in bulk, consider the following:
Typical adult hospitals require higher volumes of:
- 18G
- 20G
- 22G
Pediatric hospitals require more 22G and 24G.
Common catheter materials:
- FEP (cost-effective, firm)
- PTFE (smooth insertion)
- PU (more flexible, premium option)
Material impacts:
- Patient comfort
- Kink resistance
- Price positioning
Individual sterile blister pack
OEM private label packaging
Hospital bulk cartons
Tender-specific labeling
Private labeling is important for distributors building their own brand.
Reliable manufacturers should provide:
- CE certificate
- ISO 13485
- Sterilization validation (EO)
- Product technical datasheet
- Shelf life report (usually 3–5 years)
For large tenders:
- MOQ: 10,000–100,000 pcs
- Lead time: 20–40 days depending on order volume
- Stable supply chain for raw materials
Most manufacturers require 10,000–20,000 pcs per size. OEM packaging may increase MOQ.
Yes. Common customization includes:
- Private label branding
- Customized packaging
- Different catheter materials
- Safety mechanism selection
Usually 3–5 years under sterile packaging conditions.
In many EU markets, safety-engineered devices are recommended or required to reduce needlestick injuries.
Global healthcare demand continues to increase due to:
- Aging populations
- Expanding hospital infrastructure
- Growth of outpatient infusion therapy
- Stricter safety regulations
Hospitals are shifting toward:
- Safety IV cannulas
- High-quality catheter materials
- Reliable long-term suppliers
For distributors, securing stable manufacturing partners is becoming more important than price alone.
For hospitals, medical distributors, and government tender projects, product reliability and regulatory compliance are critical.
We supply:
- Standard IV cannulas (14G–24G)
- Safety IV cannulas
- CE & ISO certified products
- OEM private label solutions
- Bulk wholesale quantities
- Stable monthly production capacity
If you are sourcing IV cannulas for:
- Hospital procurement
- National tenders
- Medical distribution
- Private label medical brand
Contact our sales team to request:
- Product catalog
- Technical datasheets
- Sample orders
- Bulk quotation
Submit your inquiry through our contact page to receive pricing and documentation within 24–48 hours.
While the terms “IV cannula” and “IV catheter” are often used interchangeably, precise terminology matters in clinical documentation and purchasing contracts.
Understanding the technical and commercial differences helps buyers:
- Avoid specification errors
- Improve supply consistency
- Select appropriate safety features
- Optimize purchasing budgets
For distributors and hospital buyers, choosing the right IV cannula supplier is just as important as choosing the right product.